Publication: Red Grape skin and seeds
Red grape skin and seeds: extraction and characterization of flavonoids (anthocyanins) and wine production waste biomass valorization
1Santzouk Samir and Co. General Partnership, PANAX, Chrissostomou Smirnis 14, Agios Konstantinos, Aetoloacarnania, GR30100, Greece
2 FIChemE, CEng Department of Chemical Engineering, University College London, Torrington Place, London, WC1E7JE, U.K..
3Department of Environmental Engineer , University of Patras, 30100 Agrinio, Greece
Received: July11, 2019; revised:,2019
The last few years the interest in the exploitation of the winery residues is rapidly increasing. Both grape skin and seed extracts could become a valuable source of antioxidant compounds. Their valorisation towards that direction facilitates maintaining the environmental equilibrium. In the present study the method of producing an ethanolic extract of the red grape berries is presented and characteristics of it are shown.The method of the extraction process involved maceration followed by percolation; in the end of the process a strict protocol for quality control was applied. The extracts obtained via the specific extraction can be used either as individual medicinal for cures or in combination with other natural extracts to reestablish the healthy natural functions of the body. The current study, aims to production of pharmaceutical products such as capsules, tablets, drops, powder and elixirs from the red grape berries. The most important feature of this production is that all materials and products are non-toxic, natural and friendly to the environment.
Key words: red grape, extraction, flavonoids, anthocyanins.
INTRODUCTION
From its humble origins, grape production has developed into the world’s most important fresh fruit crop. Worldwide grape production in 2002 was about 62 million metric tons [1]. Approximately 66% of the production was fermented into wine [OIV, 2005]. Grape production is largely restricted to climate regions, similar to those of the indigenous range of Vitis vinifera. Grape culture is further largely restricted to regions characterized by Mediterranean-type climates [1].
The phenolic compounds in grapes (seed, skin, and pulp) can be divided into two groups, phenolic acids and flavonoids. Some flavonoids include catechin, epicatechin, procyanidins and their polymers and ester forms, quercetin, and red and blue anthocyanins. Flavonoids and related polyphenols from grape and grape seeds have generated remarkable interest based on positive reports of their antioxidant properties and ability to serve as free radical scavengers [2].
Flavonoids are natural products that are formed only in the bark or in the pulp of the fruit. The predominant flavonoids found in red grapes are anthocyanins and flavonols, which are almost exclusively present in glycosylated forms. They have antioxidant, anti-inflammatory, anti-allergic, diuretic, antibiotic and anti-carcinogenic properties. In particular, anthocyanins extracted from grape skins have been used as natural food colorants for over a century, but the compounds have recently received much attention due to their antioxidant activities and health promoting effects.

Fig.1.The biosynthetic way of phenolic compounds leading to flavonoids, lignins and the main subgroups. [3]
Grape seed extracts are a major source of anthocyanins. Anthocyanins are of particular interest to the food colorant industry due to their ability to impart vibrant colors. They have been used as traditional herbal medicines due to their diverse physiological abilities to treat conditions such as hypertension, pyrexia, liver disorders, dysentery and diarrhea, urinary problems and the common cold. The consumption of anthocyanins may play a significant role in preventing lifestyle-related diseases such as cancer, diabetes, and cardiovascular and neurological diseases [4].
Red grape seeds appear to have the highest percentage (62% w/w) of phenolic compounds [catechins, pro-anthocyanidins, concentrated tannins], while red grape bark contains significant, but lees (33%w/w) amount of phenolic compounds [anthocyanins, phenolic acids, flavonols, hydroxycinnamates].
Red grape skin and seeds have been used in order to produce a variety of pharmaceutical products [1].
In this work the medicinal properties of red grape skin and seeds (dried red grape berries) are presented; a method of valuable compounds extraction is shown; extracts characterization is demonstrated.
MATERIALS AND METHODS
Solvents.
Pure alcohol and deionized water were used as solvents.
Plant materials.
Red Grapes were obtained from Greece. They were grown on the most suitable soil; only natural fertilization was employed; throughout their production synthetic fertilizers, pesticides, insecticides, fungicides or herbicides were not used.
Plant collection.
The plant harvest was based on a strict protocol and collection rules. The harvest took place during the summer months (July-August) late in the morning to avoid moisture. After harvesting, meticulous cleaning of the red grape berries from all kinds of impurities (organic or inorganic) was done. Growth of harmful enzymes and fungi should be ruled out. Moisture is a key factor facilitating their growth. Therefore possibility of growth increases when the red grapes berries are packaged in liquid form. Thus this type of packaging was avoided. The transport length of the berries was as short as possible and it never exceeded 48 hours; the grape berries were transported in well-ventilated packages (perforated bags, baskets of reeds, boxes full of holes etc.).
Drying process.
The red grapes berries first went through a process called stabilization, via which fungi and all bacteria that can cause berries decay were naturally eliminated. The method is as follows: the grape berries were positioned in a monolayer form on top of a sieve tray, where they were sprayed by a solution of 60% pure ethanol, dispensed in a form of very fine droplets, so that a very thin layer of ethanol wetted all the external area of the berries. Ethanol was then left to evaporate.
The grape berries were dried. The drainage took place outdoors (sun or shade, it deepens) for 3 weeks.
Extraction method.
The dried grape berries were crushed in the form of a coarse powder (5 mm). The powder was mixed with the solvent, which can be water, pure alcohol or a mixture of pure alcohol and water in proportion 65:35. The crushed seeds were allowed to soak for 3 days. During this process the mixture was shaken regularly and at the end of the third day the extract had a pulp like texture. This process is called maceration.
Percolation was the subsequently selected method of extraction. The dried grape berries pulp formed via maceration was placed in a special apparatus for 4 days with sufficient amount of pure ethanol, enough to completely cover the macerated mixture. It was then placed inside the percolator, a gravity driven form of extractor, of a specific diameter designed to process a well-defined mass of the macerated material according to the equipment specifications. After this process the extract was kept for 24 hours until the supernatant liquid was clear. Finally the liquid extract was separated via gravitational filtration.
Analysis.
Quality controls were made during the procedure. In the end, the final quality control of the extract was conducted. Those control tests were based on strict protocols which involve analytical methods which have been developed in our laboratory through over 20 years of experience; they were based on traditional analytical methods of Galenea and French pharmacology but PANAX has advanced and specialized their applicability and reliability. It included determination of alcoholic degree and degree of acidity (pH) of the crude, fiber content, coefficient of expansion, bitterness limit and immediate anthocyanin identification which were used to confirm that the employed plant was red grape.
HPLC analysis.
The samples were also analyzed using a Luna, 4.6×150 mm, 5 m particle size, 100 °A pore size column. A 40:60 acetonitrile:water solution was used as an isocratic mobile phase for 15 min at 0,5 ml/min flow rate and 30 ◦C. The injection volume was 20 microliter and detection was through a UV detector set at 235 nm.
RESULTS AND DISCUSSION
The pharmacological and nutraceutical benefits derived from phenolic compounds are closely related to their antioxidant and singlet oxygen quenching ability. These phenolic compounds are able to trap and quench free radicals, and their antioxidant potentials have been shown to be four to five times that of vitamin C or E [2].
The results from the HPLC analysis of a tincture 10% dried grape berries extract are observed at figure 2. It can been seen that the extract contains a number of compounds manifested by the obvious overlapping peaks, but two of those predominate one at 2,950 min and one at 5,150 min.
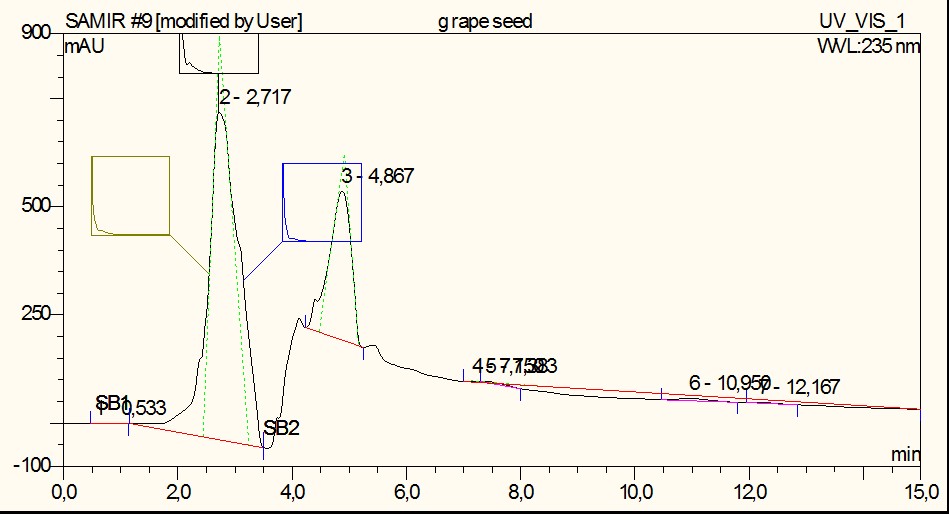
Fig. 2.HPLC absorption diagram at 235 nm, obtained from ethanolic dried grape berries extract. Absica shows retention time in min and ordinate arbitrary absorption units.

Fig. 3.Typical UV spectrum of the compound with retention time of 2.72 min of an extract from dried grape berries.
Figure 3 shows a typical UV spectrum of the peak at 2.72 min. As can be seen in Figure 3 the maximum UV absorption occurs at 215.2 and 278 nm. Further analysis for composition identification is currently underway.
In this work the treatment of dried grape berries to form ethanolic extracts. They lead to production of pharmaceutical products such as capsules, tablets, drops, powder and elixirs. The most important feature of this production is that all materials and products are non-toxic, natural and friendly to the environment.
CONCLUSION
During the wine-making processes, a lot of by-products are produced, including grape skin and seeds. Grape skin, which represents about (5–10) % of the total dry weight of the grape berry, generally contains the highest concentration of tannin [5]. In this work we presented how an ethanolic extract of red grape berries was obtained; a preliminary characterization of it via HPLC and UV is shown. The obtained extract characteristics were tested by a number of methods verifying the quality of its identity.
Acknowledgements: This project has received funding from the European Union’s Horizon 2020 research and innovation program under the Marie Sklodowska-Curie grant agreement No 778168
REFERENCES
